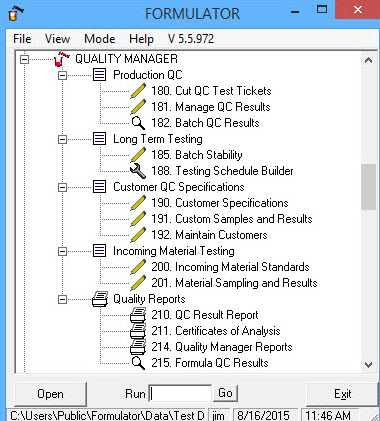
Quality Manager utilizes the Quality Control Test definition created in FORMULATOR QC Maintenance with QC Series Editor and the QC Procedures and tests created by Formula In Lab-Book Formulator Maintenance QC Maintenance tab development editor. It also provides the capability to define specifications and create Quality Test Tickets by defined batch and by customer with batch tickets or integrated with Batch Manufacturing software tied to batch tickets in integrated ERP Manufacturing software.
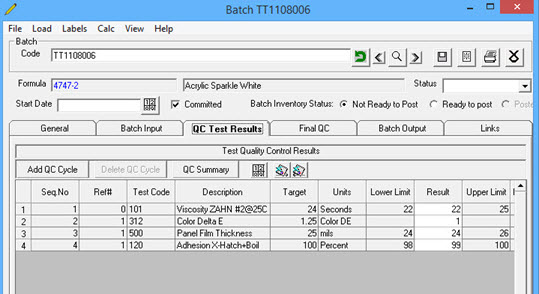
Quality Manager provides the user tools to record and document multiple QC test results and material adds, by formula batch and outputs the data to a listing for batch analysis and can create a Certificate of Analysis by batch, by product, or by customer.
Batch Quality Control allows multiple testing cycles, saving data as batch additions are made. There are no limits to the number of QC cycles and additions. As materials are added the amount of each add is documented and added to the total batch quantity. Add tickets can be generated by the QC Chemist to assure that the batch mixer makes the correct additions and that the progress of the batch is documented. The final QC results are then available to generate the basis for a batch review and Certificates of Analysis.
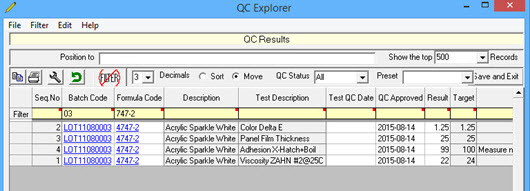
QC Explorer allows the user to review batch QC by batch by test and also enter test results into the grid.
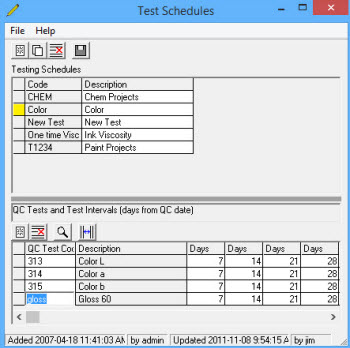
Long Term Testing is set up using the schedule builder, then will lay out the testing schedule and capture the results to archive for analysis.
PM5
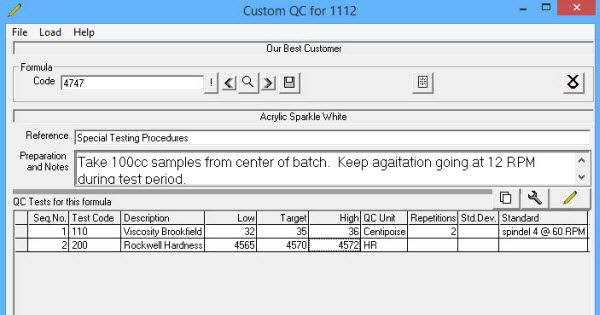
Customer QC Specifications are set up in the customer file and linked to Formulas by specified QC tests. When doing a batch or Certificate of Analysis for that customer the designated QC specifications will be used instead of the standard QC tests for the formula.
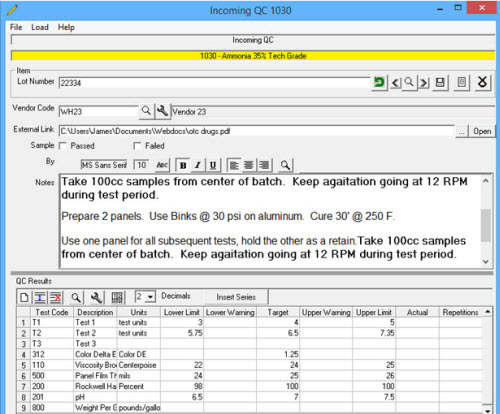
Incoming QC provides the user the tools to test incoming raw materials by lot and archive the results. Also allows linking to Vendor Certificate of Analysis supplied in electronic format (usually a pdf). Can both test to verify key requirements or perform round robin testing.
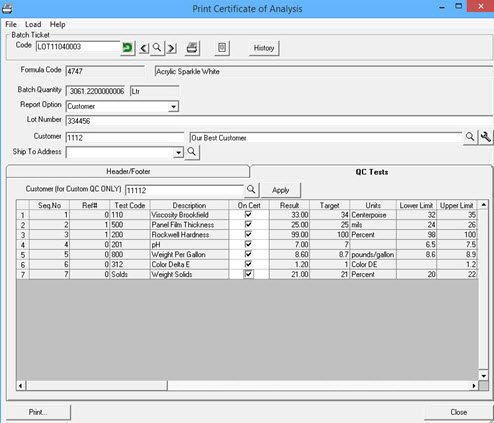
Certificates of Analysis can be created directly from the batch QC testing results. If there were multiple cycles of testing the final results by test will be output on the CofA. If the batch was for a specific customer that had alternet QC test requirements the customer’s tests will set the standard. Even if a batch is completed and posted the User can continue to test and add new test results that will be used for the CofA output. For materials that are held and later tested just as shipped additional QC tests can be entered and output on a new CofA. The CofA is a crystal report and can be saved as a pdf and electronically stored and sent to the customer or printed and mailed with shipping documents.
What is included in Quality Manager?
- Materials QC specifications by raw material, formula or by product for specific customers
- Integration with Formula QC Test Procedures
- Integration with QC Test definition and documents requirements by Formula
- Creates Quality Control Procedure and Data collection sheets by batch
- QC batch sheets can be tied to ERP manufacturing software batch ID via INTEGRATOR
- Storage of data from multiple tests repeated on the same batch and output test results for analysis
- Saves batch additions and associated tests as the batch is modified
- Provides the ability to create a new formula from the starting formula plus material additions
- Archives test result by laboratory or production batch for future review by formula by batch ID
- Certificate of Analysis output by product or by product by customer based on final approved QC test results
Why Consider Quality Manager?
- Documents and archives specifications, then saves related adds and test results
- Provides the data to identify production delays and areas for formulation improvement
- Fully ANSI SQL compatible ODBC data connectivity allows data sharing as you build your database of testing procedures and test and performance results.
- Can be used as a QC test result archive for incoming raw materials and/or standard production
- Integrates with FORMULATOR Lab-Book for QC test procedures and test requirements
- Available for Microsoft Access, Microsoft SQL Server, & MYSQL
- All of the latest calculating and data handling tools
- Latest Windows graphic tools make it easy to learn, easy to use, easy to train others
- Low cost makes it easy to own
